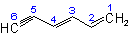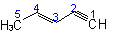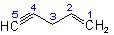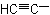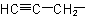IUPAC Naming of Unsaturated Compounds:
(Drag And Scroll to Animate Model)
Naming of UnSaturated Hydrocarbons.
Naming of Unsaturated Compounds
Hydrocarbons having one double bond are named by replacing the ending "-ane" of the name of the corresponding saturated hydrocarbon with the ending "-ene". If there are two or more double bonds, the ending will be "-adiene", "-atriene", etc The chain is so numbered as to give the lowest possible numbers to the double bonds.
Examples
|
1,4-Hexadiene |
|
Hydrocarbons having one triple bond are named by replacing the ending "-ane" of the name of the corresponding saturated hydrocarbon with the ending "-yne". If there are two or more triple bonds, the ending will be "-adiyne", "atriyne", etc.
The chain is so numbered as to give the lowest possible numbers to the triple bonds. Only the lower locant for a triple bond is cited in the name of a compound. Hydrocarbons having both double and triple bonds are named by replacing the ending "-ane" of the name of the corresponding saturated hydrocarbon with the ending "-enyne", "-adienyne", "-atrienyne", "-enediyne", etc .
Numbers as low as possible are given to double and triple bonds even though this may at times give "-yne" a lower number than "-ene". When there is a choice in numbering, the double bonds are given the lowest numbers.
|
1,3-Hexadien-5-yne |
|
|
3-Penten-1-yne |
|
|
1-Penten-4-yne |
|
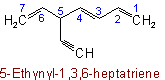
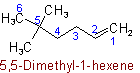
Unsaturated branched acyclic hydrocarbons are named as derivatives of the unbranched hydrocarbons which contain the maximum number of double and triple bonds.
If there are two or more chains competing for selection as the chain with the maximum number of unsaturated bonds, then the choice goes to (1) that one with the greatest number of carbon atoms; (2) the number of carbon atoms being equal, that one containing the maximum number of double bonds.
In other respects, the same principles apply as for naming saturated branched acyclic hydrocarbons. The chain is so numbered as to give the lowest possible numbers to double and triple bonds.
Examples
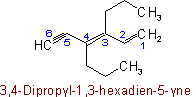
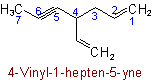
The names of univalent radicals derived from unsaturated acyclic hydrocarbons have the endings "-enyl", "-ynyl", "-dienyl", etc., the positions of the double and triple bonds being indicated where necessary. The carbon atom with the free valence is numbered as 1.
Examples
|
Ethynyl |
|
|
2-Propynyl |
|
|
1-Propenyl |
|
|
The following names are retained (for unsubstituted radical only): |
|
Vinyl (for ethenyl) |
|
|
Allyl (for 2-propenyl) |
|
|
Isopropenyl(for 1-methylvinyl) |
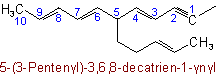
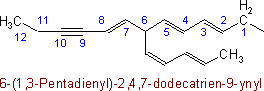
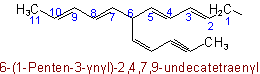
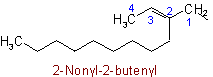
When there is a choice for the fundamental chain of a radical, that chain is selected which contains (1) the maximum number of double and triple bonds; (2) the largest number of carbon atoms; and (3) the largest number of double bonds.
Examples