Crown Ethers and Cryptands
(Drag And Scroll to Animate Model)
Crown ethers are cyclic chemical compounds that consist of a ring containing several ether groups. The most common crown ethers are cyclic oligomers of ethylene oxide, the repeating unit being ethyleneoxy, i.e., –CH2CH2O–. Important members of this series are the tetramer (n = 4), the pentamer (n = 5), and the hexamer (n = 6). The term "crown" refers to the resemblance between the structure of a crown ether bound to a cation, and a crown sitting on a person's head. The first number in a crown ether's name refers to the number of atoms in the cycle, and the second number refers to the number of those atoms that are oxygen. The name 18-crown-6 indicates that there are 18 atoms in the ring, 6 of which are oxygen. These compounds are important co-solvents. The interior of the cavity is water like, whereas the exterior is hydrocarbon like. So a metal ion inside the cavity can be "carried" into an organic solvent.
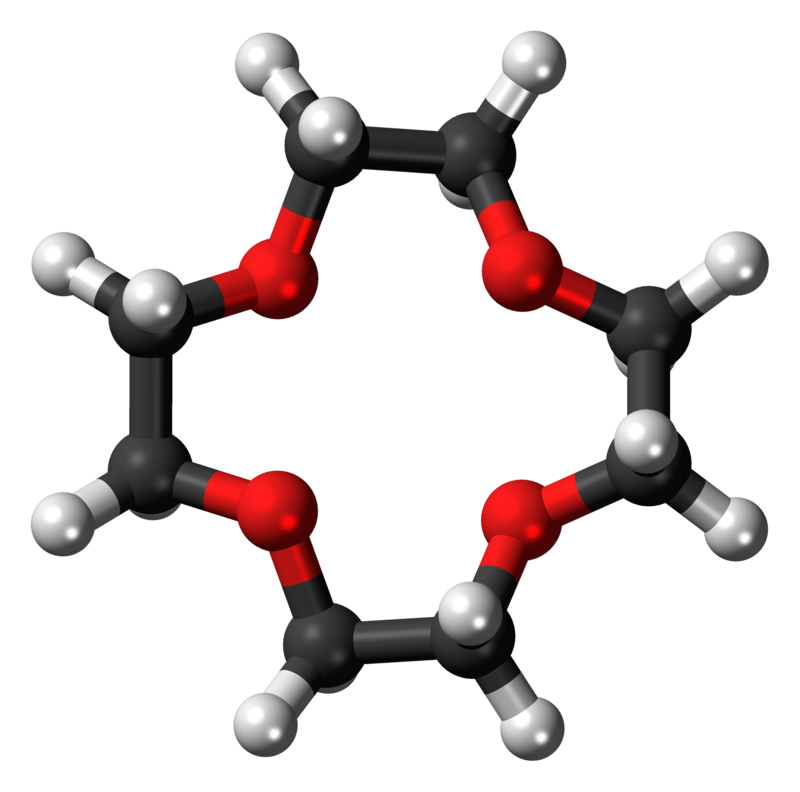
Example 1:12-crown-4
Example 1: Like other crown ethers, 12-crown-4 complexes with alkali metal cations. The cavity diameter of 1.2-1.5 Å gives it a high selectivity towards the lithium cation (ionic diameter 1.36 Å)
Open in Full Screen
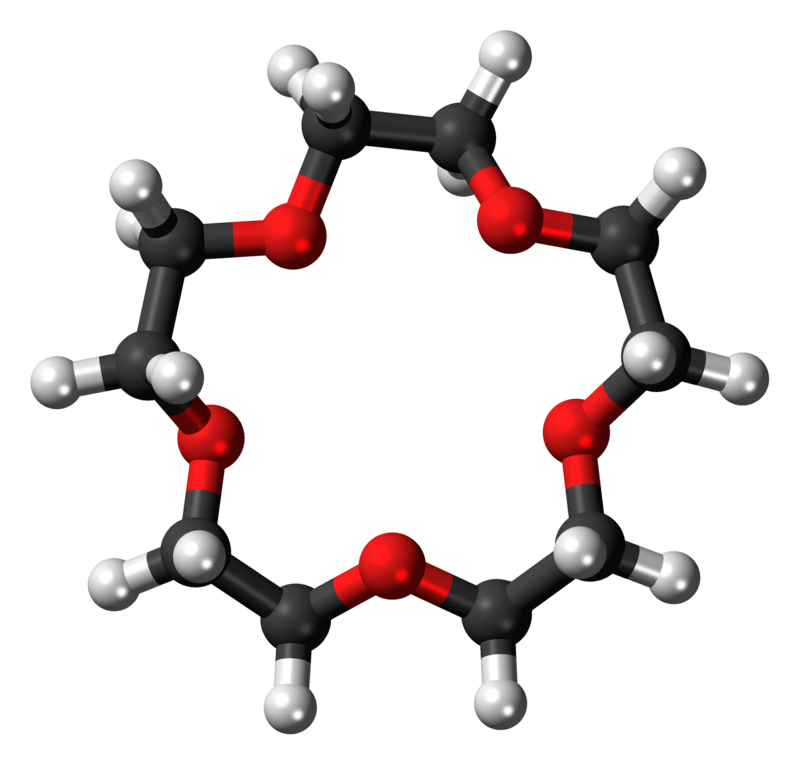
Example 2: 15-crown-5
15-Crown-5 is a crown ether with the formula (C2H4O)5. It is a cyclic pentamer of ethylene oxide that forms complex with various cations, including sodium (Na+)and potassium (K+), however, it is complementary to Na+ and thus has a higher selectivity for Na+ ions.
Open in Full Screen
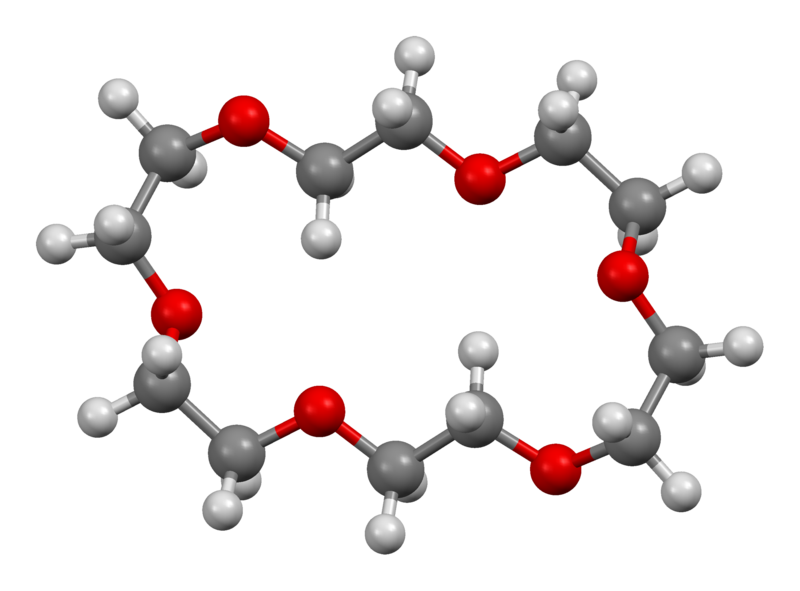
Example 3: 18crown6
18-Crown-6 is an organic compound with the formula [C2H4O]6 and the IUPAC name of 1,4,7,10,13,16-hexaoxacyclooctadecane. It is a white, hygroscopic crystalline solid with a low melting point.Like other crown ethers, 18-crown-6 functions as a ligand for some metal cations with a particular affinity for potassium cations (binding constant in methanol: 106 M−1). Dipole moment of 18-crown-6 is 2.76 ± 0.06 D in cyclohexane and 2.73 ± 0.02 in benzene.The synthesis of the crown ethers led to the awarding of the Nobel Prize in Chemistry to Charles J. Pedersen.
Open in Full Screen
Example 4:Cryptand
[2.2.2]Cryptand is the organic compound with the formula N(CH2CH2OCH2CH2OCH2CH2)3N. This bicyclic molecule is the most studied member of the cryptand family of chelating agents.It is a white solid. Their high affinity for alkali metal cations illustrates the advantages of "preorganization", a concept within the area of supramolecular chemistry. For the design and synthesis of [2.2.2]cryptand, Jean-Marie Lehn shared the Nobel Prize in Chemistry. The compound was originally prepared starting with the diacylation of the diamine-diether.

Open in Full Screen











